All authors discussed the results and commented on the manuscript. A: Given : Atomic mass of Li 6.9412 amu Isotopic mass of 6Li isotope 6. assisted with spectroscopic and chemical mapping experiments. The number of neutrons in an atom can be determined by the difference between the atomic mass and the number. Therefore, a lithium atom has three protons and three electrons. The atomic number of an element is equal to the number of protons and electrons in that element.
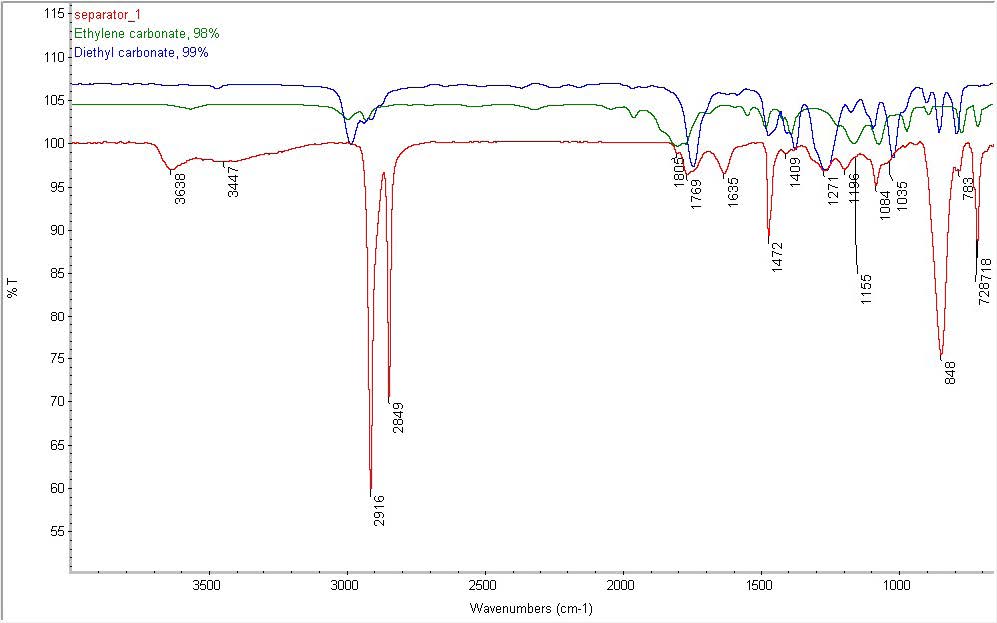
conducted electrochemical and cryo-EM characterization. Lithium is the 3rd element of the periodic table so its atomic number is 3. conceived the idea and designed the experiments. Department of Energy, under the Battery Materials Research Program and Battery 500 Consortium Programs. acknowledges support from the Assistant Secretary for Energy Efficiency and Renewable Energy, Office of Vehicle Technologies of the U.S.
acknowledges financial support by the German Research Foundation (DFG) under grant no. 3 to find Lithium on periodic table.Another way to read periodic table and locate an element is by using group number (column) and period number. Lithium was discovered by Johann Arvedson in. The lithium atom has a radius of 152 pm and a Van der Waals radius of 181 pm. The number of electrons in each of Lithium's shells is 2, 1 and its electron configuration is He 2s 1. Therefore you can directly look for atomic number. Lithium (atomic symbol: Li, atomic number: 3) is a Block S, Group 1, Period 2 element with an atomic weight of 6.94. Periodic table starts at top left ( Atomic number 1) and ends at bottom right (atomic number 118). acknowledges support from the National Defense Science and Engineering Graduate Fellowship and the Stanford Graduate Fellowship. The atomic number is usually in the top left or top right corner of the elements box, but it could be in a different spot. The atomic number increases from left to right. acknowledges the National Science Foundation Graduate Research Fellowship Program for funding. Chen for facilitating cryo-EM capabilities and A. Lithium-7 atom is the stable isotope of lithium with. Thus, since the atomic mass of iron is 55.847 amu, one mole of iron atoms would weigh 55.847 grams. There you can find the metals, semi-conductor(s), non-metal(s), inert noble gas(ses), Halogens, Lanthanoides, Actinoids (rare earth elements) and transition metals.The authors thank D. The atomic mass is useful in chemistry when it is paired with the mole concept: the atomic mass of an element, measured in amu, is the same as the mass in grams of one mole of an element. Please note that the elements do not show their natural relation towards each other as in the Periodic system. The first chemical element is Hydrogen and the last is Ununoctium.

The chemical elements ofįor chemistry students and teachers: The tabular chart on the right is arranged by Atomic number. This list contains the 118 elements of chemistry.
Separation and Concentration Purification RequestĬhemical elements listed by atomic number The elements of the periodic table sorted by atomic numberĬlick on any elements name for further chemical properties, environmental data or health effects. Atomic Data for Lithium (Li) Atomic Number 3 Atomic Weight 6.941 Reference E95 : Isotope : Mass : Abundance : Spin : Mag Moment : 6 Li: 6.015121: 7.So fluorine has more protons in its nucleus. In your case, lithium has an atomic number equal to 3 and fluorine an atomic number equal to 9. Plant Inspection & Process Optimalisation So, you know that atomic number, which represents the number of protons that can be found in the nucleus of an atom, increases as you move from left to right across a period.
